|
“And nivolumab in combination with chemotherapy represents a potential new neoadjuvant option for these patients”, he added. “CheckMate 816 is the first phase III study to show benefit of an immunotherapy plus chemo combination for resectable non-small-cell lung cancer”, said presenting author Patrick Forde, from the Johns Hopkins Kimmel Cancer Center in Baltimore, Maryland, USA, at the virtual AACR Annual Meeting 2021. MedwireNews: Among patients with operable non-small-cell lung cancer (NSCLC), supplementing neoadjuvant platinum-doublet chemotherapy with nivolumab leads to a significant boost in pathological complete response (pCR) rate, indicate trial data. doi:10.1200/JCO.2021.39.15_suppl.Author By Shreeya Nanda, Senior medwireNews Reporter Surgical outcomes from the phase 3 CheckMate 816 trial: Nivolumab (NIVO) + platinum-doublet chemotherapy (chemo) vs chemo alone as neoadjuvant treatment for patients with resectable non-small cell lung cancer (NSCLC). Neoadjuvant Opdivo (nivolumab) plus chemotherapy significantly improves pathologic complete response in patients with resectable non-small cell lung cancer in phase 3 CheckMate-816 trial.Neoadjuvant Opdivo (nivolumab) plus chemotherapy significantly improves event-free survival in patients with resectable non-small cell lung cancer in phase 3 CheckMate -816 trial.No patients died due to treatment-related AEs in the experimental arm versus 2 patients on the control therapy. Grade 3/4 surgery-related AEs were reported in 2 patients in the combination arm and no patients in the chemotherapy arm. Notably, no increase in median duration of surgery and length of hospitalization was observed between the 2 arms. Surgery was cancelled for patients who experienced disease progression or adverse effects (AEs) as well as for other reasons. Additionally, 83% of patients in the combination arm and 75% of those in the chemotherapy arm underwent definitive surgery. In total, 64% of patients have stage IIIA disease. 3 Patients were randomized to receive 360 mg of nivolumab plus chemotherapy every 3 weeks (n = 179) or the same chemotherapy backbone every 3 cycles (n = 179). Investigators enrolled patients with stage IB to IIIA resectable NSCLC with an ECOG performance status of 1 or less and no EGFR or ALK alterations.  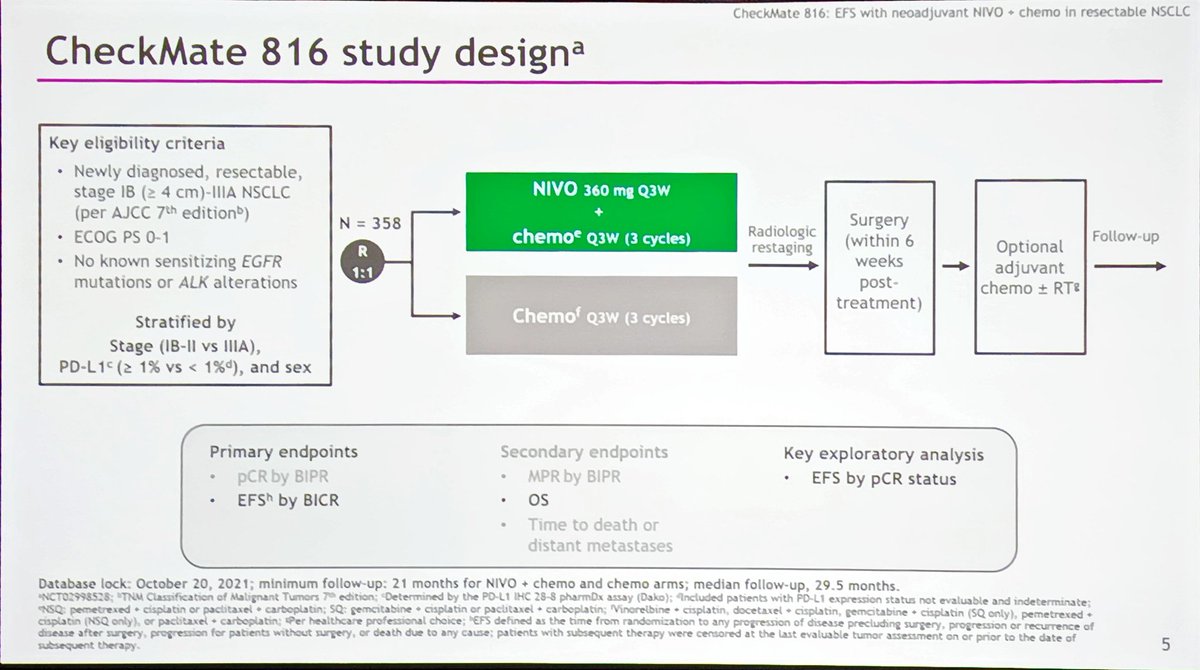
“The positive event-free survival data seen with neoadjuvant nivolumab plus chemotherapy is groundbreaking and can have important implications for how we treat resectable NSCLC.” 
“While the intent of surgery is curative in resectable NSCLC, between 30% to 55% of patients experience recurrence after surgery and ultimately succumb to the disease, presenting a strong need for additional options that can disrupt this cycle,” Nicolas Girard, MD, PhD, professor of respiratory medicine at Paris Saclay University and head of the Thorax Institute Curie Montsouris in Paris, said in a press release. 2 Results from the trial indicated that patients who received nivolumab and chemotherapy had a pCR rate of 24% vs 2.2% among those who were treated with chemotherapy alone (OR, 13.94 99% CI, 3.49-55.75 P <.0001).įull findings from CheckMate 816 will be presented at an upcoming medical conference. In addition to yielding a statistically significant improvement in EFS over chemotherapy alone, the combination has previously demonstrated a significant improvement in pathologic complete response (pCR) rate within this patient population. Treatment with neoadjuvant nivolumab (Opdivo) plus chemotherapy resulted in a significant improvement in event-free survival (EFS) among patients with resectable non–small cell lung cancer (NSCLC), according to findings from the phase 3 CheckMate 816 study (NCT02998528).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
